Note: This is an excerpt from The Ultimate Diet 2.0 that acts as a guide to the topic of calorie partitioning. First I’ll describe what calorie partitioning refers to along with examining the P-ratio.
This leads into a discussion of the hormonal changes that occur in response to both dieting and overfeeding.
Finally I’ll look at how the seemingly contradictory goals that we have in terms of changing body composition can be addressed via cyclical dieting.
Table of Contents
Calorie Partitioning
At a very fundamental level, the problem that natural bodybuilders and athletes have is one of partitioning; that is, where the calories go when you eat more of them or come from when you eat less of them.
In an ideal universe, every calorie you ate would go to muscle tissue, with none going into fat cells; you’d gain 100% muscle and no fat. In that same ideal universe, every calorie used during dieting would come from fat stores; you’d lose 100% fat and no muscle. Unfortunately, we don’t live in an ideal universe.
As I mentioned early in this book, some hapless individuals will lose as much as one pound of muscle for every 2-3 pounds of fat that they lose when they diet. Typically, those same individuals will put on about the same amount of fat and muscle when they overfeed. Thus is the balance of the universe maintained.
More genetically advantaged individuals tend to put more calories into muscle (meaning less into fat) when they overeat and pull more calories out of fat cells (and less out of muscle) when they diet. They stay naturally lean and have few problems dieting. Once again, you aren’t one of them or you wouldn’t be reading this booklet in the first place.
When talking about partitioning of calories, researchers refer to something called the P-ratio. Essentially, it represents the amount of protein that is either gained (or lost) during over (or under) feeding. So a low P-ratio when dieting would mean you used very little protein and a lot of fat.
A high P-ratio would mean that you used a lot of protein and very little fat. It looks like, for the most part, P-ratio is more or less the same for a given individual; as I mentioned above, they will gain about same amount of muscle when they overfeed as they lose when they diet. This is yet another example of the body’s attempts to maintain itself at a ‘normal’ level.
So what controls P-ratio. As depressing as this is, the majority of of the P-ratio is out of our control; it’s mostly genetic. We can control, maybe 15-20% of it with how we eat or train. Supraphysiological amounts of certain compounds (supplements) and, of course, anabolic steroids, can also affect the P-ratio.
What Determines P-Ratio?
So what are the main determinants of calorie partitioning? Obviously, hormones are crucially important. High testosterone levels tend to have positive paritioning effects (more muscle, less fat) while chronically high levels of cortisol have the opposite effect (less muscle, more fat).
Thyroid and nervous system activity affect not only metabolic rate but also fat burning; optimized thyroid and nervous system levels mean better fat burning, which means less muscle loss when you diet. It also means less fat gain when you overeat. Unfortunately, levels of these hormones are basically “set” by our genetics. The only way to change them significantly is with supplements or drugs. And really only drugs have a huge effect. Beyond that, there’s not a whole lot we can do to control them.
Another factor controlling P-ratio is insulin sensitivity which refers to how well or how poorly a given tissue responds to the hormone insulin. Now, insulin is a storage hormone, affecting nutrient storage in tissues such as liver, muscle and fat cells.
In that same ideal world, we’d have high insulin sensitivity in skeletal muscle (as this would tend to drive more calories into muscle) and poor insulin sensitivity in fat cells (making it harder to store calories there). This is especially true when you’re trying to gain muscle.
In contrast, when you diet, it’s actually better to be insulin resistant (note that two of the most effective diet drugs, GH and clenbuterol/ephedrine cause insulin resistance). By limiting the muscle’s use of glucose for fuel, you not only spare glucose for use by the brain, but you increase the muscles use of fatty acids for fuel.
In addition to hormonal advantages, it’s likely that the genetic elite have high skeletal muscle insulin sensitivity. They store tremendous amounts of calories in muscle which leaves less to go to fat cells. Their bodies also don’t have to release as much insulin in response to food intake.
In contrast, individuals with poor skeletal muscle insulin sensitivity tend to overproduce insulin, don’t store calories in muscle well (part of why they have trouble getting a pump, poor glycogen storage in muscle cells) and tend to spill calories over to fat cells more effectively.
Insulin Sensitivity
So what controls insulin sensitivity? As always, a host of factors. One is simply genetic, folks can vary 10 fold in their sensitivity to insulin. Another is diet. Diets high in carbohydrates (especially highly refined carbohydrates), saturated fats and low in fiber tend to impair insulin sensitivity. Diets with lowered carbohydrates (or less refined sources), healthier fats (fish oils, monounsaturated fats like olive oil) and higher fiber intakes invariably improve insulin sensitivity.
Another major factor is activity which influences insulin sensitivity in a number of ways. The first is that muscular contraction itself improves insulin sensitivity, facilitating glucose uptake into the cell. Glycogen depletion (remember this, it’s important) improves insulin sensitivity as well.
So what else controls the P-ratio. As it turns out, the primary predictor of P-ratio during over- and under-feeding is bodyfat percentage. The more body fat you carry, the more fat you tend to lose when you diet (meaning less muscle) and the leaner you are, the less fat you tend to lose (meaning more muscle).
The same goes in reverse: naturally lean (but NOT folks who have dieted to lean) individuals tend to gain more muscle and less fat when they overfeed and fatter individuals tend to gain more fat and less muscle when they overfeed.
The question is why. That is why is body fat percentage having such a profound impact on P-ratio. Well, there are a few easy answers. One is that body fat and insulin sensitivity tend to correlate: the fatter you get, the more insulin resistant you tend to get and the leaner you are the more insulin sensitive you tend to be.
A second is that, the fatter you are, the more fatty acids you have available for fuel. In general, when fatty acids are available in large amounts, they get used. This spares both glucose and protein. By extension, the leaner you get, the more problems you tend to have; as it gets harder to mobilize fatty acids, the body has less to use.
As I discuss in The Women’s Book, women use less protein for fuel than men and some of this is assuredly due to women’s carrying a higher average body fat percentage.
This increases the reliance on amino acids (protein) for fuel. The original Ultimate Diet advocated medium chain triglycerides (a special type of fatty acid that is used more easily for fuel than standard fats) and this can be a good strategy under certain circumstances.
But that’s not all and it turns out that body fat percentage is controlling metabolism to a much greater degree than just by providing fatty acids. Research over the past 10 years or so has identified body fat stores as an endocrine tissue in its own right, secreting various hormones and protein that have major effects on other tissues.
Perhaps the most important, and certainly the one most talked about is, leptin but that’s far from the only one. Tumor necrosis factor-alpha, the various interleukins, adiponectin and other compounds released from fat cells are sending signals to other tissues in the body which affect metabolism.
Without getting into all of the nitpicky details (many of which haven’t been worked out yet), I just want to talk a little about leptin (if you read my last book, this will all be familiar ground).
Leptin: The Short Course
Leptin is a protein released primarily from fat cells although other tissues such as muscle also contribute slightly. Leptin levels primarily correlate with body fat percentage, the more fat you have the more leptin you tend to have (note: different depots of fat, visceral versus subcutaneous, show different relationships with leptin). At any given bodyf at percentage, women typically produce 2-3 times as much leptin as men. Some of this is due to having more total body fat but much of it is due to the effect of estrogen.
In addition to being related to the amount of body fat you have, leptin levels are also related to how much you’re eating. For example, in response to dieting, leptin levels may drop by 50% within a week (or less) although you obviously haven’t lost 50% of your body fat.
After that initial rapid drop, there is a slower decrease in leptin related to the loss of body fat that is occurring. In response to overfeeding, leptin tends to rebound equally quickly. In contrast to what you might think, it looks like leptin production by fat cells is mainly determined by glucose availability (you’d think it was fat intake). So whenever you start pulling glucose out of the fat cell (dieting), leptin levels go down, when you drive glucose into fat cells, it goes up.
Basically, leptin represents two different factors: how much bodyfat you’re carrying, and how much you’re eating. That is, it acts as a signal to the rest of your body about your energy stores. I’ll come back to this in a second.
Like most hormones in the body, leptin has effects on most tissues in the body and leptin receptors have been found all over the place, in the liver, skeletal muscle, in immune cells; you name it and there are probably leptin receptors there. There are also leptin receptors in the brain but I’ll come back to that below. For now, let’s look at a few of the effects that leptin has on other tissues in the body.
In the liver, leptin tends to reduce insulin secretion from the beta-cells. In skeletal muscle, leptin promotes fat burning and tends to spare glucose (and therefore amino acid use). In fat cells, leptin may promote fat oxidation as well as making the fat cell somewhat insulin resistant. Leptin also affects immune cell function, decreasing leptin impairs the body’s ability to mount an immune response. Now you know at least part of the reason you tend to get sick more when you diet. On and on it goes.
Leptin and the Brain
Now, I want you to think back to the first couple of chapters of this book, where I talked about the evolutionary reasons it’s so hard to lose body fat. To your body, becoming too lean is a very real threat to your survival. From a physiological standpoint, that means that your body needs a way to ‘know’ how much energy you have stored.
As you may have guessed, leptin is one of the primary signals (along with many others including ghrelin, insulin, protein YY and god knows what else will turn up) that signals the brain about how much energy you have stored and how much you’re eating.
All of these hormones send an integrated signal to a part of the brain called the hypothalamus that ‘tell’ it what’s going on elsewhere in your body. This causes changes in various neurochemicals such as NPY, CRH, POMC, alpha-MSH and others, to occur. This has a variety of effects (mostly bad) on metabolic rate, hormone levels and nutrient partitioning.
So metabolic rate goes down, levels of thyroid stimulating hormone, leutinizing hormone and follicle stimulating hormone (TSH, LH and FSH respectively) go down meaning lowered levels of thyroid and testosterone, levels of growth hormone releasing hormone (GHRH) go down meaning GH output can be impaired, sympathetic nervous system activity goes down, cortisol levels go up as does hunger and appetite, etc., etc. What you end up seeing is an all purposes systems crash when you try to take body fat below a certain level.
I want to point out that falling leptin has a much larger impact on the body’s metabolism than raising leptin does (unless you’re raising it back to normal). That is, the body fights against dieting to a far greater degree than it does overfeeding.
This is why, generally speaking, it’s a lot easier to get fat than it is to get lean. Of course, there are exceptions, folks who seem to resist obesity (or weight gain altogether). Research will probably find that they are extremely sensitive to the effects of leptin, so when calories go up, they simply burn off the excess calories without getting fat.
Most of us aren’t that lucky. Rather, like insulin sensitivity discussed above, researchers will probably find that leptin sensitivity is a huge factor influencing how changes in caloric intake affect metabolism. Someone with good leptin sensitivity will tend to stay naturally lean and have an easy time dieting; folks with worse leptin sensitivity (leptin resistance) won’t.
Now, as I pointed out in my last book, injectable leptin is a pipe-dream at this point, an effective dose costing nearly $1000/day (not to mention requiring twice daily injections). Using bromocriptine or other dopamine agonists seemed to fix at least part of the problems by sending a false signal to the brain, making it think leptin levels were normal.
Recent studies that have given injectable leptin to dieters show that that fall in leptin is one of the primary signals in initiating the adaptation to dieting. However, unlike in rats, injecting leptin into humans doesn’t fix all of the problems.
This is because, in humans, there is more of an integrated response to both over and underfeeding. To make this easier to understand, let’s look at some of the major things that occur when you cut calories. To understand this better, I want to take a snapshot of what happens when you either reduce or increase calories.
The Impact of Dieting
So you start your diet, reducing carbs, calories or both. Blood glucose and insulin levels are going to be reduced. This is good, it releases the ‘block’ on fat mobilization. Additionally, catecholamine release typically goes up, further increasing fat utilization. Blood levels of fatty acids will start to increase. This is good, as it tends to promote fat burning in tissues such as liver and muscle.
This effect is facilitated if you deplete liver and muscle glycogen as glycogen depletion tends to increase the use of fatty acids for fuel. The increase in blood fatty acid levels also has the short-term effect of causing insulin resistance which, as I mentioned, is a good thing on a diet since it spares glucose and helps promote fat oxidation. So far, so good, right?
Unfortunately, those are the good effects. Along with this, a lot of bad things start to happen. Although the drop in insulin causes better fat mobilization, it causes other problems. One is that testosterone will bind to sex-hormone binding globulin (SHBG) better, lowering free testosterone levels. As well, insulin is anti-catabolic to muscle, inhibiting muscle breakdown.
The increase in cortisol that occurs with dieting enhances protein breakdown as well as stimulating the conversion of protein to glucose in the liver. Cortisol also prevents the amino acid leucine from stimulating protein synthesis. Additionally, a fall in energy state of the muscle impairs protein synthesis (although it increases fatty acid oxidation). The mechanism behind this is more detail than I want to get into here.
On top of that, high blood fatty acid levels tend to impair the uptake of T4 (inactive thyroid) into the liver. There are also changes in liver metabolism that impair the conversion of T4 to T3 (active thyroid). There is some evidence that high blood fatty acid levels causes tissues to become resistant to thyroid hormone itself (this is part of why just taking extra thyroid on a diet doesn’t fix all of the problems).
There is also a drop in nervous system output (that can occur in as little as 3-4 days after you start a diet). Along with the drop in thyroid, insulin and leptin, this explains a majority of the metabolic slowdown that occurs. The change in liver metabolism (and the reduction in insulin) also impairs the production of IGF-1 from GH.
With caloric restriction comes a drop in leptin which causes various effects on tissues such as muscle, liver and fat cells. Additionally, a hormone called ghrelin (released from the stomach and responsive to food intake) will go up. The interaction of these three hormones (and probably others) send a signal to your brain (lateral hypothalamus) that you’re not eating enough (do note that the response is not immediate, there is a lag time between the changes in all of these hormones and the body’s response).
This causes changes in the various neurochemicals such as NPY, POMC and the rest to occur, signalling further changes downstream. Levels of testosterone fall (along with the increased binding to SHBG) along with an increase in cortisol, these both tend to have a negative effect on muscle mass. In addition to the problems with conversion mentioned above, thyroid output tends to decrease over time; I already mentioned the drop in nervous system output.
All of these adaptations serve two main purposes. The first is to slow the rate of fat loss, as this will ensure your survival as long as possible. Related to that, the body tends to shut down calorically costly activities.
This includes protein synthesis, reproduction and immune function; there’s little point keeping any of these functioning when you’re starving to death. The drop in leptin, and the changes in hormones that occur are a huge part of why men tend to lose their sex drive (and ability) and women lose their period when they get lean/diet hard.
The second is to prime your body to put fat back on at an accelerate rate when calories become available again. As I mentioned earlier, this makes perfect evolutionary sense, even if it presents a huge pain in the ass to us. Ok, enough about dieting, what about overfeeding.
The Impact of Overfeeding
To a great degree, most of the adaptations that occur with dieting reverse when overfeeding. Actually, that depends a lot on the situation. As I mentioned above, the body as a whole tends to defend against underfeeding better than it does against overfeeding.
This is why it’s generally easier to gain weight than to lose it. Studies where leptin has been increased above normal (i.e. to try and cause weight in overweight individuals) have generally borne this out: except at massive doses, raising leptin above normal does very little.
There are a few reasons for this. One theory is that normal leptin levels send essentially a 100% signal, that is they tell the body that all systems are normal. It should seem clear that raising leptin above 100% isn’t going to do much. Another problem is something I alluded to above: leptin resistance.
It’s thought that people have varying degrees of leptin resistance which means, in essence, that they don’t response as well to leptin as they should. On top of this, when leptin levels go up, it appears to stimulate resistance to itself. That is, when leptin gets and stays high, it causes you to become resistant to its effects.
Both theories make good evolutionary sense. Your body doesn’t want to be lean but it doesn’t really mind getting fat. If calories are available all the time, it would make little sense for you to get full and/or start burning them off. This is what would happen if you were extremely sensitive to leptin. So high levels of leptin induce resistance to itself; keeping you hungry and eating while the food is available.
Note: There is also the simple fact that the modern environment tends to drive food consumption through what is called the non-homeostatic or hedonic system. Humans don’t just eat to maintain the body’s fuel stores or energy levels. We eat because food tastes good and it’s clear that tasty food can overwhelm any biological homeostatic system that exists.
But we’re not really talking about raising leptin above normal here, we’re talking about reversing or preventing the drop that occurs with dieting. In that situation, many of the above adaptations will reverse to one degree or another (depending on how lean you are, how long you diet, and how long you overeat).
So now you increase your calories and carbs. This raises blood glucose and insulin, reversing the binding of testosterone to SHBG; cortisol also goes down. With increased carbohydrates, you increase both liver and muscle glycogen. In the muscle, while this decreases fat oxidation, this improves protein synthesis (along with the increase in insulin and testosterone and the decrease in cortisol).
Of course, with increasing insulin, there is a decrease in blood fatty acid concentrations which improves insulin sensitivity. Skeletal muscle insulin sensitivity is enhanced even more by exercise.
The decrease in blood fatty acids, along with changes in liver metabolism will improve both the uptake and conversion of T4 to T3; along with improvements in nervous system output, this will help to increase metabolism.
And, of course, there are all of the central adaptations that occured during dieting, that will reverse to some degree while overfeeding. Leptin will go up (noting that it goes up more quickly than bodyfat comes on) along with insulin, ghrelin goes down. This signals the hypothalamus that you’re eating again, and many of those changes will reverse. So NPY, CRH, POMC and the rest go back towards normal, helping to renormalize all of the hormones that were screwed up in the first place.
To a very limited degree, some of these adaptations would be expected to try and limit fat gain and, to a very limited degree, this is what happens. But, as above, the body is better at preventing weight loss than weight gain.
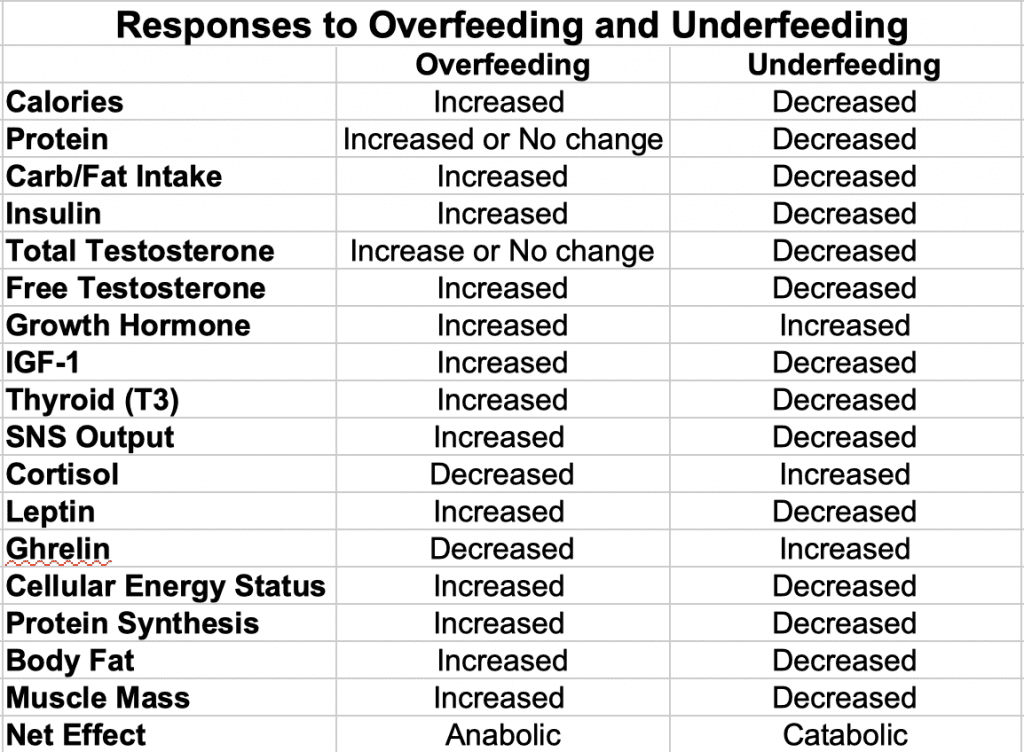
Summary of the Changes in Hormones
Ok, let’s put the above two sections together in chart form so that it’s easier to see.
So now, in greater detail, you’re starting to understand the problems involved, especially for the genetically normal. Underfeeding is necessary for fat loss but will always have a negative impact on muscle mass.
It also induces any number of adaptations that tend to prevent further fat loss. Overfeeding is necessary to gain but will always have a negative impact on fat mass. However, it can reverse many (if not all) of the adaptations that occur with dieting.
A Final Note on Leptin
Hopefully the above sections have made you realize that there is far more to the adaptations to either dieting or overfeeding than just leptin. Rather, it’s an integrated response involving leptin, insulin, ghrelin, fatty acids, liver, fat cell and skeletal muscle adaptations, and probably factors that haven’t been discovered yet. This probably explains why injecting leptin into dieting humans only reverses some but not all of the adaptations.
For example, just injecting leptin would be expected to fix a defect in TSH (and thyroid output) but it won’t fix the problems with conversion that occur at the liver. Similarly, while injecting leptin would normalize LH and FSH output, it won’t correct the problem with increased binding of testosterone caused by lowered insulin. Hopefully you get the picture. So, you ask, what’s the solution?
The Solution: Cyclical Dieting
Ok, great, I’ve just spent nearly 10 pages making a case for cycling dieting, periods where you alternate a low-calorie intake with a higher calorie intake. in this fashion, you alternate between periods of low calories/carbs with high calories/carbs to alternate between periods of anabolism (tissue building) and catabolism (tissue breakdown). Fundamentally, of course, this is nothing new even if research has only now started to look at it and find that it works.
Several years ago, when I first started making some of the connections with leptin and everything else, this really pointed out the need to do periodic refeeds (or cheat days or whatever you want to call them) on a diet.
If nothing else, it pointed to another reason why the Bodyopus diet worked as well as it did: by force feeding carbs and calories for 2 days, not only did you refill muscle glycogen and hopefully generate an anabolic response, you probably reversed some of the adaptations inherent to dieting.
In those years, various approaches have come and gone. In general, short refeeds, lasting from 5 to 24 hours were used every so often while dieting. I’ve tried them all. The Bodyopus diet was aimed at this goal, alternating 5 days of low-cal/ketogenic dieting with 2 days of high-carbohydrate eating.
There hav been many others approaches that were similar in nature, that alternated periods of higher and lower calories. Even basic carbohydrate/calorie cycling as used by bodybuilders falls into that schema.
The question is whether or not those programs were optimal. In my opinion, they aren’t for several reasons.
One of the factors I’ve been considering to a great degree has to do with the length of the overfeeding period. While it’s true that 5 (or 12 or 24) hours of concentrated overfeeding will raise leptin, the more important question is whether that’s sufficient to ‘tell’ the brain that you’re fed. While data (especially in humans) is nonexistent, my hunch is no.
There’s a lag time of several days between the drop in leptin and the drop in metabolic rate (nervous system output) for example; I’d be surprised if a mere 12 or 24 hours was sufficient to reverse this.
Rather, I’d expect it to take a similar amount of time for the reversal to occur. The reasons I feel this way are sort of beyond the scope of this book, send me an email if you really must know.
Now, this isn’t to say that short carb-loads/refeeds aren’t of benefit. They refill glycogen, turn off catabolism and maybe induce an anabolic response to boot. They also let you eat some of the crap you’re really craving which helps psychologically.
But I doubt they are sufficient to affect metabolism very much. Instead, a longer refeed is necessary. The drawback, of course, is that longer refeeds have a tendency to put too much bodyfat back on which goes agains the entire goal of dieting.
Another problem with many cyclical dieting approaches is that they don’t coordinate training with the diet. Bodyopus was an exception but, for various reasons, I think the Bodyopus workout plan was screwy.
If anything it was backwards, putting tension workouts on low-calorie/low-carb days (where you aren’t very anabolic) and glycogen depletion workouts before you are eating a lot seemed wrong to me years ago and wronger to me now. This will make more sense as you read the next chapters.
Ultimately, all of this introductory stuff, brings us to the final question: how do we optimize the entire system to maximize fat loss and either muscle maintenance or muscle gain (or, if you’re a performance athlete, how do we generate fat loss while maintaining performance). To understand that, I need to get into a few more details regarding muscle gain and fat loss, which will help you to understand the overall system.
Those questions led me to develop The Ultimate Diet 2.0, a program that carefully integrated all of the data on calorie partitioning in terms of diet, training, supplements, etc. to create an optimized schema to achieve everything (and more) described in this article.
Similar Posts:
- Insulin Resistance and Fat Loss
- The Impact of Bodyfat Percentage on Body Composition Changes
- Insulin Sensitivity and Fat Loss
- Insulin Levels and Fat Loss
- What Is The Ketogenic Ratio?

The link to enova oil is https://www.enovaoil.com/
Very nice site!
Lyle,
You said
“In contrast, when you diet, it’s actually better to be insulin resistant (note that two of the most effective diet drugs, GH and clenbuterol/ephedrine cause insulin resistance). By limiting the muscle’s use of glucose for fuel, you not only spare glucose for use by the brain, but you increase the muscles use of fatty acids for fuel.”
IF you are insulin resistant, then insulin levels/blood glucose will be high, which would not enable to fat to be used a fuel source, correct?], since insulin levels determine if fat is used as a fuel source.
Insulin resistance under calorically/carb restricted conditions is different than under hypercaloric/carb sufficient conditions. So no.
Man, Lyle, you have great articles but sometimes they are such a downer!
Sadly, Chip, how we would like life to be and how life actually is are often not the same thing. In fact, they are usually quite different.
Heya Lyle,
insensitivity being a major problem here, how about simply increasing receptor sensitivity once more?
Mark
Insulin-producing beta cells are in the pancreas, not in the liver.
Brilliant article – this website is a tremendous resource, thank you sincerely. I recently read your article on insulin – which was also brilliantly helpful. When you mention that “more highly refined” carbohydrates can contribute to issues with insulin sensitivity in the context of higher sat fat and reduced fiber content in the diet, is it impossible for more highly refined carbohydrates to have a similar impact in the context of a diet with appropriate sat fat and fiber content? I have had trouble losing fat (yet have gained muscle) whilst trialling a more flexible approach to my carbohydrate choices, despite eating the same number of calories whilst eating “clean”, and am trying to iron out my misconceptions about food-hormone responses… Could I be insulin sensitive?
First, huge thanks for sharing your insights as I’m working steadily toward my goals. My question -I think- is about energy partitioning. I started a caloric deficit (to about 1400-1500/day) about 4 weeks ago and have dropped 7 lbs of pure fat (as assessed by scale and 7-site skin fold test). Specifically, weight dropped from 168-162 lbs and body fat percentage from 16.9% to 12.8% (5’11, weight train 3 days a week). Does this mean that I have a high P-ratio and can I expect to lose more LBM as I get to lower body fat percentages? Furthermore, as I reverse the diet and aim to put on mass, should I expect to gain a high percentage of body fat relative to LBM.
First, huge thanks for sharing your insights as I’m working steadily toward my goals. My question -I think- is about energy partitioning. I started a caloric deficit (to about 1400-1500/day) about 4 weeks ago and have dropped 7 lbs of pure fat (as assessed by scale and 7-site skin fold test). Specifically, weight dropped from 168-162 lbs and body fat percentage from 16.9% to 12.8% (5’11, weight train 3 days a week). Does this mean that I have a high P-ratio and can I expect to lose more LBM as I get to lower body fat percentages? Furthermore, as I reverse the diet and aim to put on mass, should I expect to gain a correspondingly high proportion of body fat relative to LBM?
William, please read
https://www.bodyrecomposition.com/muscle-gain/initial-body-fat-and-body-composition-changes.html
As BF%age gets lower, there is a change in all of this. What is easy to do from 18% to 12% is much harder from 12% to 8%
Darren: Of course it’s not impossible to lose fat with what you’re describing. I was the one who first wrote about flexible dieting and clearly it works.