So I’ve written a lot about protein requirements for athletes in my career. It’s a topic I’ve followed with some interest for decades, I wrote an entire book on the topic in 2008 or so. Well guess what I’m talking about again today. Hint: it’s protein requirements for athletes.
Table of Contents
Protein Requirements for Athletes
The most recent piece I wrote on the topic addressed a current idea in field where maximal per meal protein intake is about 0.25 g/lb (about 0.55 g/kg). This is based on research showing that this amount of protein maximizes MUSCULAR PROTEIN SYNTHESIS. This is assumed to represent the optimal protein requirement for growth.
Related: How Much Protein Do I Need to Grow?
In the original piece, I gave a number of criticisms of that idea including the following:
- Most studies concluding this use whey protein rather than whole food. This not only alters digestion and appearance kinetics but the food matrix is important as the presence of other nutrients and compounds impacts on how foods work in the body.
- Many of the training protocols are debatable as to their relevance to how serious athletes or bodybuilders train as they tend to use a handful of sets tops.
- The studies invariably ignore muscle protein breakdown, primarily due it being hard to measure (note: this isn’t a good reason to ignore an important biological process)
- Muscle protein synthesis is an idiotic endpoint to use to begin with. I’ll address this point again in detail.
I’d note that some individuals, well one specific individual (not named in the article) was unhappy with my piece. They even went so far as to write a rebuttal in the AARR. I’ll come back to that. So let’s get to it.
Muscle Protein Synthesis is an Idiotic Endpoint
There are several reasons I think that setting protein requirements based solely on muscle protein synthesis responses is idiotic.
One is that it ignores muscle protein breakdown completely. Net protein balance in any tissue is the, err, balance between the two. So for protein we can define:
Net Protein Balance = Protein Synthesis – Protein Breakdown
From the standpoint of increasing muscle mass, that means that we could conceivably alter either. We could increase MPS or decrease MPB or both. While it’s debated now, we have known for years that amino acids and insulin have differential effects in this regard. Here’s a neat graphic I drew forever ago showing how this works.
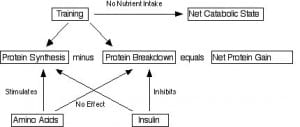
And while many have made a big issue that MPB is a relatively “small” part of muscle growth that doesn’t mean it should be ignored in my opinion. It’s also about 30% or so of the overall effect and, well, I don’t consider that small.
Note: another argument regarding MPB is that it may be bad to decrease it due to its importance in helping with tissue remodeling. Perhaps another article for another day. In any case, MPB is primarily ignored due to being methodologically difficult (and expensive) to measure. This is a dumb reason to ignore something.
More Protein Means More Anabolism
To that I’d add that Wolfe’s group showed quite clearly that more protein was more anabolic in the body and a lot of that was mediated by decreased protein breakdown. They also used whole foods (beef patties) rather than isolated whey protein.
Now, they were measuring whole-body protein balance. Eating more protein didn’t have an impact on muscle protein synthesis and I’m not saying that it did. It just affected the rest of the body. There is also emerging evidence that excess protein (some of which is burned for energy) is stored within the gut in the short-term.
This probably occurs so that the body can release those aminos into the bloodstream when you haven’t eaten, providing them for other tissues in the body. Hmmm…It’s always been said that the body can’t store protein but this is also looking like it’s not right either.
Lean Body Mass is More than Muscle
The bigger issue is one so simple that I am baffled at the ability of supposed experts in this field to not understand it. We divide the body into Lean Body Mass (LBM) and Fat Mass (FM). In a leanish male athlete, FM might be 10-15% leaving 85-90% LBM. But what is LBM?
Many think of LBM as being muscle mass but that’s not true. Quite in fact, LBM makes up, on average, about 45% of total LBM (it can range from about 40-50%). That means that, on average 55% (40-60%) of your LBM is NOT muscle.
So what is it?
Well it’s everything else that’s not fat. This includes thing like water, minerals and glycogen which aren’t relevant here. Importantly it includes your organ mass, most of which are made of some amount of protein and which go undergo daily protein turnover. This process is not 100% efficient meaning that some amino acids will be lost and have to be replaced.
Fun fact: during long-term protein malnutrition, once LBM gets below a certain point, the body will break down organ mass to get amino acids. Then you die.
You can’t simply ignore the protein requirement of those tissues. Well you can and people clearly do but it’s stupid to do so.
Add to that things like peptide hormones that are made out of amino acids. Add to that things like connective tissues such as tendons and ligaments that contain amino acids (i.e. the focus on glycine for tendon health).
I think you get the idea.
Protein Requirements For Athletes
Adding to that, not only do those tissue utilize and require protein but some of them assuredly adapt to long-term training in athletes. In fact, we know that they do over time. To suggest that they don’t have an amino acid requirement on top of MPS is idiotic. It’s myopic as hell.
Of course it’s generally been felt that the turnover rate of those other tissues was very slow and that might argue that their day-to-day protein requirements are too slow to worry about.
Let’s find out if this idea still holds.
Protein Synthesis Rates in Non-Muscle Tissues
To address the topic again, I want to look at a recent(ish) article titled Protein synthesis rates of muscle, tendon, ligament, cartilage, and bone tissue in vivo in humans.
The paper set out to examine exactly what the title stays: the rate of protein synthesis in muscle AND a bunch of other tissues. Towards that goal it took 6 subjects who were slated for knee surgery.
Yes, this is a small sample size. Yes, the study needs to be replicated. Preferably by an independent lab (science rests on INDEPENDENT replication).
But this is not uncommon where you have access to a population group in a research setting where you can do this. The subjects were already there for surgery and getting cut into. Why not use that situation to study something?
Lyle Has Done Science
On that note, a research group (Whipp and Ward) I interned with in college (that’s right Layne and James and Brad, I have “done science” you ad hominem dropping jackasses) at UCLA did studies on what are called Carotid Body Resected (CBR) patients. The carotid body is an oxygen sensor and drives respiration when O2 levels are low.
And UCLA had the only large population of CBR patients since they did the surgery there. So the researchers studied them to see what would happen during a VO2 max test. I helped with lab setup, exercise tests and running the data analysis on the computers. Anyhow, the sample size wasn’t big. It was just the one that existed and which were clustered into an area to do the research.
Same thing with this knee surgery study.
Back to the Study
I won’t even attempt to explain the methodology as it’s super complicated and beyond my paygrade. Basically they did a radiolabelled amino acid infusion, measured a bunch of stuff, did a bunch of calculations and came up with the numbers. I’ll take at face value that they did it all right since I don’t have the background to assess it.
This is what they found:
Using stable isotope methodology, we showed that average basal protein synthesis rates of various musculoskeletal tissues are within the same range of skeletal muscle protein synthesis rates, with fractional muscle, tendon, bone, cartilage, ligament, menisci, fat, and synovium protein synthesis rates ranging between 0.02 and 0.13% per hour in vivo in humans.
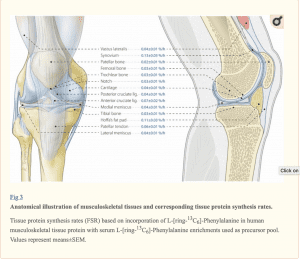
Which, translated means that, at rest, all of the tissues measured had identical protein synthetic rates. Muscle, the connective tissues, fat tissue and everything else. Here is the image showing what and where the specific tissues measured are along with the protein synthesis rates.
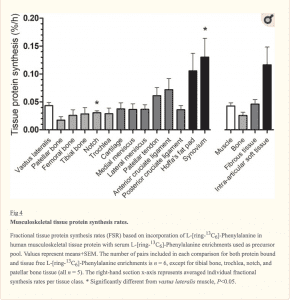
Since that’s a little bit difficult to see, here is an image showing the actual rates of protein synthesis. While there are visual differences, they didn’t reach statistical significance in most cases.
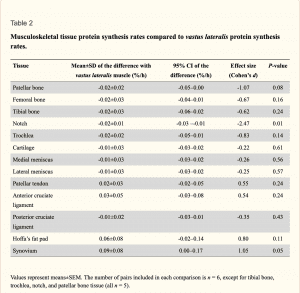
Finally here is a chart showing the differences in protein synthesis rate from skeletal muscle. Note that all values are tiny and none reached a statistically significant (P<0.05) level. In a statistical sense, all values were identical. Or rather, they weren’t different.
So what this suggests is that many other protein containing tissues, including connective tissues, have the same basal protein synthesis rates as skeletal muscle. Which means that they have a protein requirement.
This raises the question of whether or not those tissues adapt or regenerate, which might also impact on their protein requirements. As they write:
Knowledge of basal musculoskeletal tissue protein synthesis rates enables us to further explore the capacity of these tissues to regenerate. Skeletal muscle plasticity is well appreciated since muscle tissue has shown to be highly responsive to both anabolic and catabolic stimuli.
To assess whether other musculoskeletal tissues are capable of displaying some degree of plasticity, more work is required to address the impact of various factors on tissue protein synthesis rates.
Like good scientists, they acknowledge that more work is needed. Of course, the results of this study need to be replicated to ensure that it’s an accurate finding.
At the same time, there is limited research showing that connective tissues respond to a training stimulus and that providing amino acids at the right time helps with this. They state:
It has previously been observed that gelatin supplementation can stimulate collagen synthesis following exercise [55], and collagen hydrolysate supplementation has been reported to increase collagen content in the knee of osteoarthritis patients [61], and may decrease knee pain in athletes with activity-related joint pain[62].
From a clinical perspective this is more than interesting, because identifying which specific proteins or protein fractions are responsive to external stimuli may enable us to develop more effective therapies in treating and/or preventing injuries.
I discussed some of that in my book on Optimal Nutrition for Injury Recovery, written after I broke my leg.
To that I’d add that this is clearly something that must be considered in terms of protein requirements for athletes. Long-term adaptations to tendons, ligament, bone, etc. is all important for long-term improvements in performance. And protein is an important aspect of those adaptations.
As I wrote in The Protein Book over a decade ago
In addition to all of the body’s uses of protein described above, there are a number of processes of extra importance to athletes. This includes the repair and replacement of damaged proteins, remodeling of the proteins within muscle, bone, tendon and ligaments, maintenance of optimal functioning of all of the metabolic pathways that use amino acids (presumably these pathways are up regulated in athletes due to training), supporting lean body mass gains, supporting immune system function, and possibly others (4).
As you’ll see in Chapter 4, this makes determination of protein requirements for athletes quite difficult as it is unclear how much additional protein might be required to optimally support all of the processed listed above.
At the time it was more speculative but I always enjoy being a decade ahead of the game. In 2020, I’d say more and more emerging evidence supports the idea.
As the researchers conclude:
In conclusion, basal fractional muscle, tendon, bone, cartilage, ligament and menisci protein synthesis rates range between 0.02 and 0.13% per hour in vivo in humans. Fractional tissue protein synthesis rates of tendon, bone, cartilage, ligament and menisci do not differ substantially from muscle tissue protein synthesis rates, suggesting that these musculoskeletal tissues may express a greater level of tissue plasticity than generally believed.
So there ya’ go. Tissues other than skeletal muscle have a protein turnover rate identical to skeletal muscle. It clearly adapts to training over time. The conclusion from this is very clear so far as I’m concerned:
Protein requirements for athletes must take this into account.
Do we need more research on this? Absolutely. Do we need to quantify how much extra protein is required for these tissues to optimally adapt from training? Absolutely. But that’s not really my point here.
The point is that, as I originally argued, using muscle protein synthesis (MPS) as the singular endpoint to set protein requirements is idiotic. It ignores muscle protein breakdown, it ignores roughly 50-60% of the body’s other LBM, some of which requires protein on a day-to-day basis.
It ignores other tissues that clearly adapt over time with training. Tissues that may adapt more quickly than we thought. It is based on generally unrealistic or at least non-representative training programs and it’s invariably based on isolated whey protein instead of whole food.
And none of that paragraph represents some insanely theoretical or esoteric argument. It’s really basic physiology and metabolism. Basic physiology and metabolism that a certain individual I did not name originally but am about to seems unaware of.
Jorn Trommelen and Stu Phillips
So as I said in the introduction, when I wrote my original piece, it upset someone. I didn’t name him originally but will now, it’s Jorn Trommelen. In contrast to what I stated in the video, Jorn does not actually work in Stu’s lab. Instead he works with Luc Van Loon. Which still doesn’t change how wrong they all are about this.
Now Jorn (and Stu) and I have history, no shock. They were both in my group but both left. Stu was incapable of understanding that HIIT in beginners is a mistaken recommendation due to the effort involved. He, like Jorn, was also not happy with my criticism of their initial protein feeding at night study.
I pointed out it had three independent variables (total protein intake, inadequate amounts in one group, timing) which is just shit poor science. And it’s not as if it couldn’t have been fixed easily by simply giving the control group a protein drink to have at any time other than bedtime. Boom, matched protein, sufficient protein in both groups and you’re looking only at timing. You know, good science.
But among his other warbling, Stu tried to defend the issue of inadequate protein intake (it was 1.2 g/kg) in the one group, I cited ONE OF HIS OWN papers on protein requirements suggesting that 1.6 g/kg was optimal.
To which he responded “It was only a guess.” which is just sad. Basically to defend a shitty new paper, he decided to just dismiss ONE OF HIS OWN papers out of hand. I said so and he left which was just fine.
In another moment of rocket sciencedom has recently said something to the effect of “If testosterone is so important to building muscle, how do women build muscle?” which shows an utter ignorance of the physiology involved. Primarily he’s confusing absolute and relative levels and it’s clear that even changes in testosterone within a woman’s normal range impacts on her muscle growth (I discuss this in detail in my Birth Control and Athletic Performance e-book).
A physiology I daresay I understand better than he does. Ahem:
Well Jorn was involved in all of this, in fact he’s a co-author on the piece of crap protein timing study. After I pointed out my clear criticisms of it, he wrote me a wall of text explaining how I didn’t understand science.
Ah the old “You don’t understand science” ad-hominem. Used by so many bullshit artists such as Layne Norton and Brad Schoenfeld to dismiss real criticism out of hand. I think during that wall of text he accused me of an ad hominem attack. You know, as he wrote one that didn’t address a single point of my criticism. Isn’t it ironic?
Except that it’s funny because I sure seem to understand science better than he does if he can’t understand why having three independent variables is shitty research just like I understand that not-blinding research is sloppy methodology and introduces bias….
But he would leave my group shortly thereafter. And seems to still be pretty butthurt over me. I mean, for goodness sake, I went out of my way NOT to name him in my original article since I didn’t need to. And he still got all wound up over it. He should consider some good collagen protein to thicken his skin. So should a lot of people in this industry.
The AARR “Rebuttal”
Now in the AARR he made a few different arguments. Among them….
He asserted that Wolf’es work on protein intakes and whole body protein metabolism was done with poor methodology. It’s a standard dismissal when a lab gets a different result than you. Well ok. Take that up with Wolfe, not me.
Another was that in his and Stu’s lab they have the most accurate tools to measure muscle protein synthesis. Even if that’s true, so what? You can have the best telescope in the world and it won’t help IF YOU’RE LOOKING AT THE WRONG THING.
And simply, they are. They are myopically focused on the one thing: the impact of dietary protein on muscle protein synthesis. Everything else is ignored which is just nonsensical. But when you only look at the one thing you only see the one thing. Jorn would appear to need a refresher on protein metabolism. In fact, since I’m a giver, I’ll send him a copy of my Protein Book via email (message me through the contact page, buddy). God knows he could use it if his understanding is this superficial.
As of June 2020, I have not heard from Jorn so I guess he didn’t want his free book.
Edit: As of September 2020, I have not heard from Jorn so I guess he didn’t want a free book.
Protein Requirements for Athletes Redux Redux
And that’s that. This paper provides even more support for my original thesis: focusing on muscle protein synthesis (MPS) alone is nonsensical. To beat a dead horse, muscle is only 40-50% of LBM leaving 50-60% non muscle. Some of that other LBM requires protein.
Including connective tissues.
Which have the same basal rate of protein turnover as skeletal muscle.
And which clearly adapt in response to training.
To ignore the protein requirements for those tissues, or the rest of the body’s LBM is myopically dumb even if two “top tier” protein researchers can’t seem to see why it’s dumb. That a bunch of people in the fitness industry keep repeating it without even thinking about it critically raises other questions to me but that’d be another entire article.



Facebook Comments